Neonatal diarrhoea in calves (NCD) has been, is, and will remain a major pathological concern in breeding cattle for the foreseeable future (Herman et al., 2022). This is in spite of research over decades, the development of vaccines against enterotoxigenic E. coli, Rotavirus, Coronavirus and constant improvement in the optimisation of the transfer of passive immunity.
Although modern cattle farming has made great improvements in herd management, animal facilities and hygiene, calf diarrhoea is still problematic due to the multi-factorial nature of the disease and the vulnerability of the newborn calf.
The prevalence of pathogen and disease incidence can vary by geographical location of the farm, farm management practices and herd size. That said, these four major enteric pathogens are involved in NCD worldwide:
- Rotavirus
- Coronavirus
- E. coli
- Cryptosporidium parvum
Is antimicrobial use justified in the treatment of NCD?
Neonatal diarrhoea has a significant impact on the profitability of the farm and on animal welfare. Besides the administration of rehydration remedies (e.g., Immustart®, Dialyte, Hydra F) which are essential for supportive treatment, early diagnosis and rational use of antimicrobials can attenuate clinical disease, decrease the release of pathogens to the environment, and decrease animal mortality (Izzo et al., 2015; Naylor, 2006).
The administration of antimicrobials is further justified when considering animal welfare from the public point of view, and increased productivity from the livestock producer's point of view. Antimicrobials should only be used according to guidelines in place for responsible antibiotic use.
When should antimicrobials be considered?
Etiological diagnosis is now possible on farm thanks to rapid tests such as HuveCheck®. These tests will direct the veterinarian to using an antibiotic in the presence of E. coli or an antiprotozoal in the presence of Cryptosporidium.
Bacterial enteritis
Many studies have shown microbial proliferation during diarrhoea (Gomez and Weese, 2017; Jang et al., 2019), and in particular, the development of E. coli in the small intestine. E. coli F5 is the most frequently detected causative agent of NCD, causing profuse watery diarrhoea but without the loss of intestinal epithelium integrity (Foster and Smith, 2009).
Antibiotic choice and route of administration
For an antibiotic to be effective, its concentration vis-à-vis the bacterium at the infectious site (blood or intestine) must be higher than its MIC (minimal inhibitory concentration).
There is little data on the distribution of antibiotics in the intestine following parenteral administration. Only fluoroquinolones and ceftiofur which provide sufficient serum and intestinal concentrations (Foster et al., 2016). However, following international recommendations from the World Health Organisation (WHO) and World Organisation for Animal Health (WOAH), 3rd and 4th generation cephalosporins and fluoroquinolones are considered critically important antimicrobials and their use should be avoided in veterinary medicine.
If an antibiotic is injected during septicaemia, it seems thus reasonable to add oral treatment to obtain effective blood and digestive concentration.
Aminoglycosides such as apramycin (Apravet®) and paromomycin (Parofor®) are bactericidal antibiotics which are very poorly absorbed in the digestive tract which therefore results in high concentrations (above the MIC) in situ after oral administration. Aminoglycosides are concentration-dependent antibiotics. Recent studies support high doses with a short duration to avoid adaptive resistance.
From a study conducted in 2015 (Figure 1), paromomycin concentrations in the lumen greatly exceeded the MIC 90 value of 1024 µg/g (Paromomycin MIC range: 2 > 1024, Vet Path IV project, strain collection 2015-2016) after oral administration at a dose of 50 mg/kg body weight (Fichant, 2015).
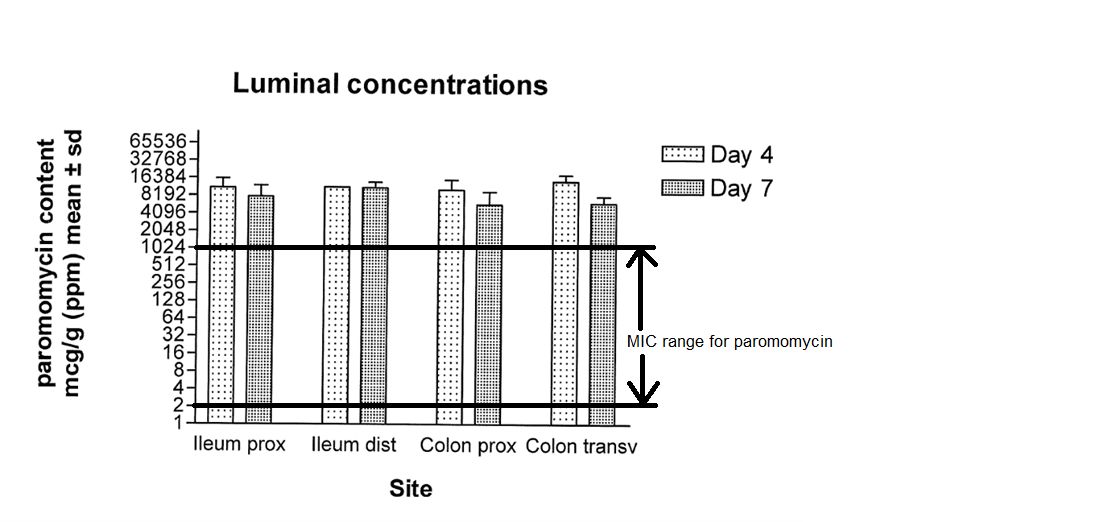
It is clear from the study that paromomycin concentrations exceed the MIC 90 value and that the high concentrations in situ support the use of aminoglycosides to target bacteria such as E. coli.
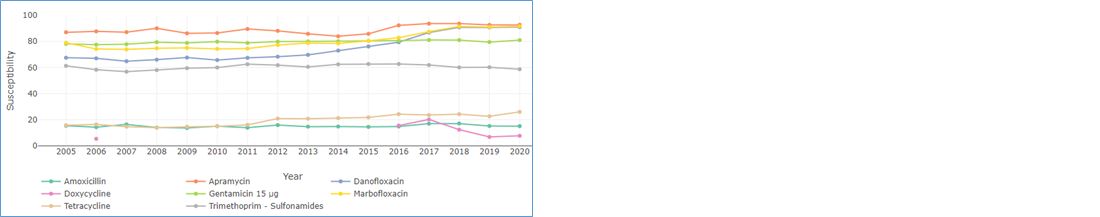
Furthermore, given the current legislation and sensitivity profiles of E. coli isolated from digestive pathologies (Figure 2) in calves, aminoglycosides should be given priority for oral use.
From a US study published in 2004 (Constable, 2004), only orally administered apramycin and marbofloxacin were found to be effective in field-observed diarrhoea. At the time of the trial, in France (Figure 2), 87% of E. coli were susceptible to apramycin and, in 2020, 91% were susceptible.
Practical administration of Parofor® and Apravet®
For a rational oral use in the treatment of neonatal calf diarrhoea, administer in milk or milk replacer as follows:
Parofor® - 50 mg paromomycin sulphate per kg body weight / day for 5 days
or
Apravet® - 40,000 IU apramycin sulphate per kg body weight / day for 5 days
In conclusion, following etiological diagnosis, antibiotics remain a rational choice in the treatment of neonatal calf diarrhoea to restore calf health and welfare and to prevent the spreading of disease. Aminoglycosides such as Parofor® and Apravet® reach sufficiently high concentrations at the requisite site and their sensitivity profiles support their use.
References are available on request


